Carbon, Nitrogen, and Water Cycles
CARBON CYCLE
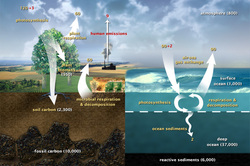
1. How does carbon exist in the atmosphere?
- Carbon moves into the atmosphere from decomposition of organic matter,
respiration by living organisms, combustion, volcanic activity, burning fossil
fuels, degassing of waterbodies, etc.
2. How are fossil fuels created? Explain.
- Fossil fuels were formed from plants and animals that lived up to about 300
million years ago. The Sun passed solar energy to the plants, and
plants were eaten by animals. When the plants and animals died and started
decaying, some of them got buried deep under the ground for many millions of
years, where they turned into fossil fuels like coal, gas and crude oil. There are three basic categories of fossil fuels: coal, petroleum (or crude oil), and natural gas.
3. Describe two ways that carbon enters the atmosphere.
- From the natural carbon cycle out of the oceans, soil, and animals, and from the burning of fossil fuels.
4. How are the oceans involved in the carbon cycle?
- The oceans influence the climate by absorbing and storing the carbon dioxide.
5. How is the temperature of the Earth partly controlled by carbon?
- Too much of it in the atmosphere and the sun's heat gets trapped and global
average temperature rises. Humans also emit carbon dioxide, primarily by burning
fossil fuels such as coal, oil and natural gas. As a result, drives global average temperatures up (which is commonly known as Global Warming).
6. What role do rocks have within the carbon cycle?
- After many years, as rocks are eroded, gases trapped in them will be released into the atmosphere so in a way rocks are "storage rooms" for gases just as our Earth is.
7. Where are you starting within the carbon cycle?
- The movement of carbon, in its many forms, between the biosphere, atmosphere,
oceans, and geospheres.
8. How much of the atmosphere is made of carbon dioxide (CO2)?
- 0.04%
9. By how much has CO2 increased in the atmosphere during the past 150
years?
- By 30%
10. Next stop = Land Plants. What did you learn?
- Carbon can go back into the atmosphere through respiration. Photosynthesis used to make nutrition for plants. The more CO2 in the atmosphere, the faster rate the plants can grow.
11. Next stop = Surface Water. What did you learn?
- 90 Gigatons of carbon is contain in the surface ocean per year approximately.
12. The deep ocean holds more than 65% of the Earth's Carbon. How much carbon does the surface ocean absorb from the atmosphere per year?
- 90 Gigatons per yr.
13. True or False: When plants die and decay, they bring carbon into
soil.
- True
14. Next stop = Marine Life. What did you learn?
- High levels of carbon in ocean waters are harmful to marine organisms, but they
need carbon to go through photosynthesis and sustain life.
15. Next stop = Deep Ocean. What did you learn?
- Carbon stays in the deep ocean for hundreds of years before continuing the carbon cycle. It holds more than 65% of Earth’s carbon.
16. When carbon enters the deep ocean, how long does it stay there?
- Carbon stays for hundreds of years once it enters the deep ocean.
True or False: Phytoplankton are tiny plants and algae that float in the ocean and take up carbon dioxide as they grow.
- True
True or False: Plants both absorb CO2 from the atmosphere and release it into the atmosphere.
- True
- Carbon moves into the atmosphere from decomposition of organic matter,
respiration by living organisms, combustion, volcanic activity, burning fossil
fuels, degassing of waterbodies, etc.
2. How are fossil fuels created? Explain.
- Fossil fuels were formed from plants and animals that lived up to about 300
million years ago. The Sun passed solar energy to the plants, and
plants were eaten by animals. When the plants and animals died and started
decaying, some of them got buried deep under the ground for many millions of
years, where they turned into fossil fuels like coal, gas and crude oil. There are three basic categories of fossil fuels: coal, petroleum (or crude oil), and natural gas.
3. Describe two ways that carbon enters the atmosphere.
- From the natural carbon cycle out of the oceans, soil, and animals, and from the burning of fossil fuels.
4. How are the oceans involved in the carbon cycle?
- The oceans influence the climate by absorbing and storing the carbon dioxide.
5. How is the temperature of the Earth partly controlled by carbon?
- Too much of it in the atmosphere and the sun's heat gets trapped and global
average temperature rises. Humans also emit carbon dioxide, primarily by burning
fossil fuels such as coal, oil and natural gas. As a result, drives global average temperatures up (which is commonly known as Global Warming).
6. What role do rocks have within the carbon cycle?
- After many years, as rocks are eroded, gases trapped in them will be released into the atmosphere so in a way rocks are "storage rooms" for gases just as our Earth is.
7. Where are you starting within the carbon cycle?
- The movement of carbon, in its many forms, between the biosphere, atmosphere,
oceans, and geospheres.
8. How much of the atmosphere is made of carbon dioxide (CO2)?
- 0.04%
9. By how much has CO2 increased in the atmosphere during the past 150
years?
- By 30%
10. Next stop = Land Plants. What did you learn?
- Carbon can go back into the atmosphere through respiration. Photosynthesis used to make nutrition for plants. The more CO2 in the atmosphere, the faster rate the plants can grow.
11. Next stop = Surface Water. What did you learn?
- 90 Gigatons of carbon is contain in the surface ocean per year approximately.
12. The deep ocean holds more than 65% of the Earth's Carbon. How much carbon does the surface ocean absorb from the atmosphere per year?
- 90 Gigatons per yr.
13. True or False: When plants die and decay, they bring carbon into
soil.
- True
14. Next stop = Marine Life. What did you learn?
- High levels of carbon in ocean waters are harmful to marine organisms, but they
need carbon to go through photosynthesis and sustain life.
15. Next stop = Deep Ocean. What did you learn?
- Carbon stays in the deep ocean for hundreds of years before continuing the carbon cycle. It holds more than 65% of Earth’s carbon.
16. When carbon enters the deep ocean, how long does it stay there?
- Carbon stays for hundreds of years once it enters the deep ocean.
True or False: Phytoplankton are tiny plants and algae that float in the ocean and take up carbon dioxide as they grow.
- True
True or False: Plants both absorb CO2 from the atmosphere and release it into the atmosphere.
- True
NITROGEN CYCLE
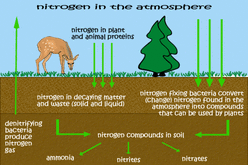
1. What are the two conditions under which nitrogen will react with oxygen? (In other words, what is necessary for nitrogen in the air to combine with oxygen?)
- Nitrogen will react with oxygen in conditions with really high temperatures and pressures. This might happen during lightning.
Nitrogen will also combine with oxygen in combustion reactions. For example, this might happen when we’re burning fossil fuel in power plants or in vehicle internal combustion engines.
2. What are the two compounds that are formed when nitrogen combines with oxygen?
- NO and NO2
3. How does nitric acid (HNO3) form?
- Nitric acid forms when NO2 reacts with water.
4. Why is nitric acid (HNO3) important?
- Nitric acid can be used by plants to obtain the nitrogen they need.
5. What percentage of the air we breathe is nitrogen?
- The air we breathe is 79% nitrogen.
6. Even though considerable nitrogen is available in the air, most plants do not use the nitrogen (N2) found in the air. Why not?
- Plants are not able to use nitrogen in this form.
7. In what compounds can plants use nitrogen?
- Plants can only use nitrogen when it has been converted or “fixed” into one of these compounds: NO3, NH3, or (NH2)2CO
8. How do animals get the nitrogen they need?
- Animals can only get nitrogen by eating plants or eating other animals (who got their nitrogen by eating plants).
9. Atmospheric nitrogen (N2) is pretty inert. This means that it does not easily break apart. When molecules do not break apart easily, it is difficult (or impossible) for organisms to use them as a nutrient source.
As a result, nitrogen fixation is the term used to describe the process of breaking up N2.
10. What is atmospheric fixation?
- In atmospheric fixation, the unusable nitrogen found in the atmosphere is converted into a form useable by plants. That process occurs under incredibly high temperatures (during lightning). The newly created compounds reach earth/soil by dissolving in water. This creates only a small percentage (5-8%) of all the nitrogen useable by plants.
11. What is industrial fixation? [This is how artificial fertilizers are made.]
- In industrial fixation, nitrogen is converted to a useable form by a chemical reaction that takes nitrogen from the atmosphere and hydrogen from a fossil fuel in high heat to form nitrogen compounds that plants can use.
12. What is biological fixation? (In your answer, describe the types of plants associated with the symbiotic relationship.)
- In biological fixation, a living organism (nitrogen-fixing bacteria) are able to convert atmospheric nitrogen into a nitrogen-containing compound that plants can use. These nitrogen-fixing bacteria live in a mutualistic relationship with legumes (bean-type plants)
13. Why is nitrogen needed by plants and animals?
- Nitrogen is necessary for plants and animals because it helps produce amino acids, proteins and nucleic acids – these help cells function correctly and make up our DNA.
- Nitrogen will react with oxygen in conditions with really high temperatures and pressures. This might happen during lightning.
Nitrogen will also combine with oxygen in combustion reactions. For example, this might happen when we’re burning fossil fuel in power plants or in vehicle internal combustion engines.
2. What are the two compounds that are formed when nitrogen combines with oxygen?
- NO and NO2
3. How does nitric acid (HNO3) form?
- Nitric acid forms when NO2 reacts with water.
4. Why is nitric acid (HNO3) important?
- Nitric acid can be used by plants to obtain the nitrogen they need.
5. What percentage of the air we breathe is nitrogen?
- The air we breathe is 79% nitrogen.
6. Even though considerable nitrogen is available in the air, most plants do not use the nitrogen (N2) found in the air. Why not?
- Plants are not able to use nitrogen in this form.
7. In what compounds can plants use nitrogen?
- Plants can only use nitrogen when it has been converted or “fixed” into one of these compounds: NO3, NH3, or (NH2)2CO
8. How do animals get the nitrogen they need?
- Animals can only get nitrogen by eating plants or eating other animals (who got their nitrogen by eating plants).
9. Atmospheric nitrogen (N2) is pretty inert. This means that it does not easily break apart. When molecules do not break apart easily, it is difficult (or impossible) for organisms to use them as a nutrient source.
As a result, nitrogen fixation is the term used to describe the process of breaking up N2.
10. What is atmospheric fixation?
- In atmospheric fixation, the unusable nitrogen found in the atmosphere is converted into a form useable by plants. That process occurs under incredibly high temperatures (during lightning). The newly created compounds reach earth/soil by dissolving in water. This creates only a small percentage (5-8%) of all the nitrogen useable by plants.
11. What is industrial fixation? [This is how artificial fertilizers are made.]
- In industrial fixation, nitrogen is converted to a useable form by a chemical reaction that takes nitrogen from the atmosphere and hydrogen from a fossil fuel in high heat to form nitrogen compounds that plants can use.
12. What is biological fixation? (In your answer, describe the types of plants associated with the symbiotic relationship.)
- In biological fixation, a living organism (nitrogen-fixing bacteria) are able to convert atmospheric nitrogen into a nitrogen-containing compound that plants can use. These nitrogen-fixing bacteria live in a mutualistic relationship with legumes (bean-type plants)
13. Why is nitrogen needed by plants and animals?
- Nitrogen is necessary for plants and animals because it helps produce amino acids, proteins and nucleic acids – these help cells function correctly and make up our DNA.
WATER CYCLE
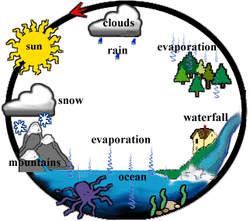
1. Define "water cycle".
- A complex process, which provides humans water to drink, oceans to sustain marine organisms, and grow crops.
2. What fraction of the Earth’s surface is covered in water?
- 3/4 of the Earth’s surface is covered in water.
3. What percentage of all the Earth’s water is in a form that is useable to humans
and land animals?
- 1%
4. Evaporation is the process where a liquid changes from its liquid state to a gaseous state. Why is evaporated water so clean?
- When water is being evaporated, the impurities in the water are left behind.
Condensation occurs when a gaseous is changed into a liquid.
Condensation is the opposite of evaporation.
When the temperature and atmospheric pressure are right, the small droplets of water in clouds form larger droplets and precipitation occurs.
5. Define transpiration:
- Transpiration is the process of evaporation through plant leaves. The plants would absorb water from the soil, and the water travels around the whole plants through the roots, eventually some of the water gets evaporated from the leaves, which adds water vapor in the air.
- A complex process, which provides humans water to drink, oceans to sustain marine organisms, and grow crops.
2. What fraction of the Earth’s surface is covered in water?
- 3/4 of the Earth’s surface is covered in water.
3. What percentage of all the Earth’s water is in a form that is useable to humans
and land animals?
- 1%
4. Evaporation is the process where a liquid changes from its liquid state to a gaseous state. Why is evaporated water so clean?
- When water is being evaporated, the impurities in the water are left behind.
Condensation occurs when a gaseous is changed into a liquid.
Condensation is the opposite of evaporation.
When the temperature and atmospheric pressure are right, the small droplets of water in clouds form larger droplets and precipitation occurs.
5. Define transpiration:
- Transpiration is the process of evaporation through plant leaves. The plants would absorb water from the soil, and the water travels around the whole plants through the roots, eventually some of the water gets evaporated from the leaves, which adds water vapor in the air.
