The Danger of Ocean Acidification
|
- Burning fossils fuels against the background of natural variability.
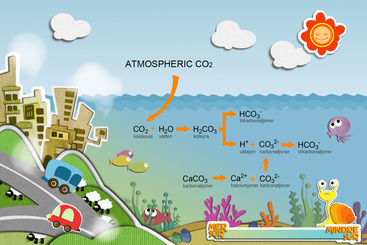
- 40% of C02 derived from fossil fuel remains in the atmosphere, the rest is taken up by vegetation on land or by the ocean, currently in about equal proportion. - The atmospheric concentration of C02 was approximately constant for several 1000 years and then began to grow rapidly with onset industrialization. - Absorbed C02 forms carbonic acid in seawater, lowering the prevailing pH level and changing the balance of carbonate and bicarbonate ions. - The decline in pH thus threatens a variety of organisms, including corals, which provide one of the richest habitats on Earth. |
- Within a century, the surface of the Southern Ocean will become corrosive to the shells of tiny snails that form a key link in the marine food chain within this highly productive zone.
- CO2 combines with water to form carbonic acid H2C03, the same weak acid found in carbonated beverages. Like all acids, this one releases hydrogen ions ( H+) into solution, in this case leaving both carbonate ions HCO3-1 and to a lesser extent carbonate ions ( CO3-2).
- Much of CO2 released into the atmosphere was destined to remain there.
- A substantial fraction would end up in the sea.
- When C02 is too high, shells and sea organisms dissolved.
- In its solid form, CO2 is used in fire extinguishers, in laboratories, and in theater and stage productions as dry ice to make fog
- Carbon dioxide is not normally considered a pollutant because it is a normal constituent of air. However, excess of carbon dioxide is considered a [pollutant because it leads to adverse effects on the environment. The higher concentration of carbon dioxide in the atmosphere is likely to increase the temperature of the atmosphere. As already discussed carbon dioxide permits the short wavelength visible radiations to pass through it but traps the longer wavelength infra-red radiations (heat waves) reflected by the earth’s surface. This trapping of heat waves causes excessive heating of earth’s atmosphere. This heating effect on earth produced in this way is called GREEN HOUSE EFFECT. The excessive heating of earth and its atmosphere can have adverse effect on our climate, which will affect all the living beings. The climate will become gradually hot.
Exposure to CO2 can produce a variety of health effects. These may include headaches, dizziness, restlessness, a tingling or pins or needles feeling, difficulty breathing, sweating, tiredness, increased heart rate, elevated blood pressure, coma, asphyxia, and convulsions. The levels of CO2 in the air and potential health problems are:
* 250 - 350 ppm – background (normal) outdoor air level
So what?
- Being aware of ocean acidification is important because if nothing is done now soon there will be no sea life. Marine animals can't live with such low pH values.
What if?
- The pH levels continue to drop? Then calcifying organisms will die off since the acidification is destroying their protection.
Says who?
- Geochemist Roger Revelle.
What does this remind me of?
- This remind me of The Lorax film, because they both talk about how human activities impact on environment.
- CO2 combines with water to form carbonic acid H2C03, the same weak acid found in carbonated beverages. Like all acids, this one releases hydrogen ions ( H+) into solution, in this case leaving both carbonate ions HCO3-1 and to a lesser extent carbonate ions ( CO3-2).
- Much of CO2 released into the atmosphere was destined to remain there.
- A substantial fraction would end up in the sea.
- When C02 is too high, shells and sea organisms dissolved.
- In its solid form, CO2 is used in fire extinguishers, in laboratories, and in theater and stage productions as dry ice to make fog
- Carbon dioxide is not normally considered a pollutant because it is a normal constituent of air. However, excess of carbon dioxide is considered a [pollutant because it leads to adverse effects on the environment. The higher concentration of carbon dioxide in the atmosphere is likely to increase the temperature of the atmosphere. As already discussed carbon dioxide permits the short wavelength visible radiations to pass through it but traps the longer wavelength infra-red radiations (heat waves) reflected by the earth’s surface. This trapping of heat waves causes excessive heating of earth’s atmosphere. This heating effect on earth produced in this way is called GREEN HOUSE EFFECT. The excessive heating of earth and its atmosphere can have adverse effect on our climate, which will affect all the living beings. The climate will become gradually hot.
Exposure to CO2 can produce a variety of health effects. These may include headaches, dizziness, restlessness, a tingling or pins or needles feeling, difficulty breathing, sweating, tiredness, increased heart rate, elevated blood pressure, coma, asphyxia, and convulsions. The levels of CO2 in the air and potential health problems are:
* 250 - 350 ppm – background (normal) outdoor air level
- 350- 1,000 ppm - typical level found in occupied spaces with good air exchange.
- 1,000 – 2,000 ppm - level associated with complaints of drowsiness and poor air.
- 2,000 – 5,000 ppm – level associated with headaches, sleepiness, and stagnant, stale, stuffy air. Poor concentration, loss of attention, increased heart rate and slight nausea may also be present.
- >5,000 ppm – this indicates unusual air conditions where high levels of other gases could also be present. Toxicity or oxygen deprivation could occur. This is the permissible exposure limit for daily workplace exposures.
- >40,000 ppm - this level is immediately harmful due to oxygen deprivation.
So what?
- Being aware of ocean acidification is important because if nothing is done now soon there will be no sea life. Marine animals can't live with such low pH values.
What if?
- The pH levels continue to drop? Then calcifying organisms will die off since the acidification is destroying their protection.
Says who?
- Geochemist Roger Revelle.
What does this remind me of?
- This remind me of The Lorax film, because they both talk about how human activities impact on environment.